Ecologists identify potential new sources of Ebola and other filoviruses
An international team of ecologists has identified the bat species with the greatest potential to harbor filoviruses-a family that includes Ebola virus.
An international team of ecologists has identified the bat species with the greatest potential to harbor filoviruses-a family that includes Ebola virus.
By Beth Gavrilles
Sexual transmission of the Ebola virus could have a major impact on the dynamics of the disease, potentially reigniting an outbreak that has been contained by public health interventions, according to research by University of Georgia ecologists just published in the Royal Society journal Biology Letters.
The potential for sexual transmission is high for three to four months after the virus has been cleared from the bloodstream, and possible for an average of seven months.
The research was prompted by the publication of data showing that viable Ebola virus remained in the semen of disease survivors for months after it was no longer detectable in their blood—and by a study reporting at least one instance of sexual transmission of Ebola.
“We realized that this could be a hidden source of the virus,” said senior author Andrew Park, an associate professor in the UGA Odum School of Ecology and College of Veterinary Medicine’s department of infectious diseases. “We wanted to find out what role sexual transmission might play in the dynamics of an outbreak.”
The researchers developed a mathematical model to test various outbreak scenarios.
First, they created a model population of 1,000 individuals and introduced Ebola virus to track its spread via regular transmission. Based on the experience of the 2014 outbreak, they assumed that many actions would be taken, from individual behavior changes to public health interventions, to control the outbreak. In the parameterized model, this resulted in one in four individuals infected throughout the population.
“An important point to make is that without the behavior changes and interventions, it would have been more like 80 percent of individuals infected,” said Park. “The actions taken in West Africa were very effective.”
Next, they set out to determine the impact that sexual transmission could have.
“We wanted to know what it would mean in terms of the size of an outbreak, how long an outbreak lasts, how likely an outbreak is to occur and the reproductive ratio of the parasite, a measure of how effectively the parasite transmits in populations,” said the study’s lead author John Vinson, a doctoral student in the Odum School.
There were, however, two components of sexual transmission about which very little is known. The first is what proportion of people who survive Ebola are actually able to transmit the virus through sexual contact; the second is how the rate of sexual transmission compares to that of regular transmission.
To overcome this lack of data, they ran the model using values that varied widely for both questions but within plausible limits.
Their results showed a clear impact from sexual transmission. When the values of both parameters—the number of sexually infectious individuals and the rate of transmission—were low, outbreaks were smaller and ended more quickly, but as the values increased, so did the size and duration of outbreaks.
The model shows that even the smallest and shortest outbreaks in the presence of sexual transmission were larger and longer-lasting than outbreaks where no sexual transmission occurred.
“Whenever we had die-outs of the directly transmitted infectious individuals, which would otherwise have spelled the end of the outbreak, we had reignition from the sexually infectious individuals transmitting the virus to the susceptible people left in the population, who then served as a source of direct transmission,” Vinson said. “Thinking about it from the parasite’s point of view, the parasite is able to persist in that population even without the direct contact transmission by symptomatic individuals.”
Park said the findings point to the importance of considering alternative pathways of disease transmission.
“There’s an increasing awareness that sexual transmission can happen in addition to a more clearly evidenced transmission route and not just with Ebola,” he said. “But there’s been very little written about how it works or what it means for the metrics that the public health community uses. Our model is ultimately translatable to other disease systems and shows how it can be done even under uncertainty of key parameters.”
The study, “The potential for sexual transmission to compromise control of Ebola virus outbreaks,” is available online at http://dx.doi.org/10.1098/rsbl.2015.1079.
Co-authors are John Drake, an associate professor in the Odum School, and Pejman Rohani, a professor in the Odum School and College of Veterinary Medicine. Support for the research was provided by the National Institute of General Medical Sciences of the National Institutes of Health under award number U01GM110744 and by the National Science Foundation.
May 10, 2016
The University of Georgia has entered into a collaborative research agreement with GeoVax Labs, Inc. to develop and test a vaccine to prevent infection from the emerging Zika virus.
The collaboration will combine the vaccine-development expertise of UGA researchers, led by Ted Ross, director of UGA’s Center for Vaccines and Immunology, with GeoVax’s novel vaccine-platform technology. Ross, a professor and Georgia Research Alliance Eminent Scholar in Infectious Diseases at the College of Veterinary Medicine, joined UGA last fall.
The mosquito-transmitted Zika virus is linked with birth defects in thousands of babies in Brazil and elsewhere; and with Guillain- Barré syndrome, a disorder in which the body’s immune system attacks the nervous system. The virus is expected to spread to most of the Western Hemisphere, with Gulf Coast states bearing the brunt of Zika outbreaks in North America.
There is no proven vaccine or treatment for Zika—closely related to the yellow fever, dengue, and West Nile viruses, which also are transmitted to people by mosquitoes.
“We believe that the expertise of our researchers combined with GeoVax’s vaccine platform can accelerate the development and testing of a vaccine for this fast-spreading viral disease,” Ross said.
Ross’s research group specializes in designing, developing and testing vaccines—including what are called VLP-based vaccines—for viruses like those noted above as well as Ebola, influenza, respiratory syncytial and HIV/AIDS.
VLPs—“virus-like particles”—mimic a live virus but do not contain genetic material; although they cannot cause infection, they elicit a strong immune response in the cells of the person being vaccinated.
Ross explained that vaccines made with VLPs give the immune system a head start in preventing infection. “When a person vaccinated with a VLP is infected by the real virus,” he said, “the immune system is ready to fight back.”
Vaccines using VLPs on today’s market are used to prevent hepatitis and papillomavirus infections, and others are in development.
GeoVax’s vaccine-platform technology takes a different approach with VLPs. Instead of introducing them in the vaccine, it uses recombinant DNA or recombinant viruses to produce VLPs in the person being vaccinated. In that way, they more closely resemble the virus generated in a person’s body during a natural infection.
The Atlanta-based company’s VLP platform has so far been used in vaccines against HIV and hemorrhagic fever viruses, including Ebola, Marburg and Lassa. The HIV vaccine has been proven safe in human clinical trials involving over 500 individuals. It also is being evaluated for use in cancer vaccines. “We believe our vaccine platform is uniquely suited to apply to the Zika virus,” said Robert McNally, GeoVax president and CEO.
Ross’s lab will test Zika VLP vaccines developed by GeoVax, as well as any emanating from his own research team.
In addition to Ross, UGA researchers working on Zika vaccines will include Ralph Tripp, Georgia Research Alliance Chair in Vaccine and Therapeutic Development; Biao He, Georgia Research Alliance Distinguished Investigator; Fred C. Davison, Distinguished University Chair in Veterinary Medicine; and Mark Tompkins, associate professor of infectious diseases.
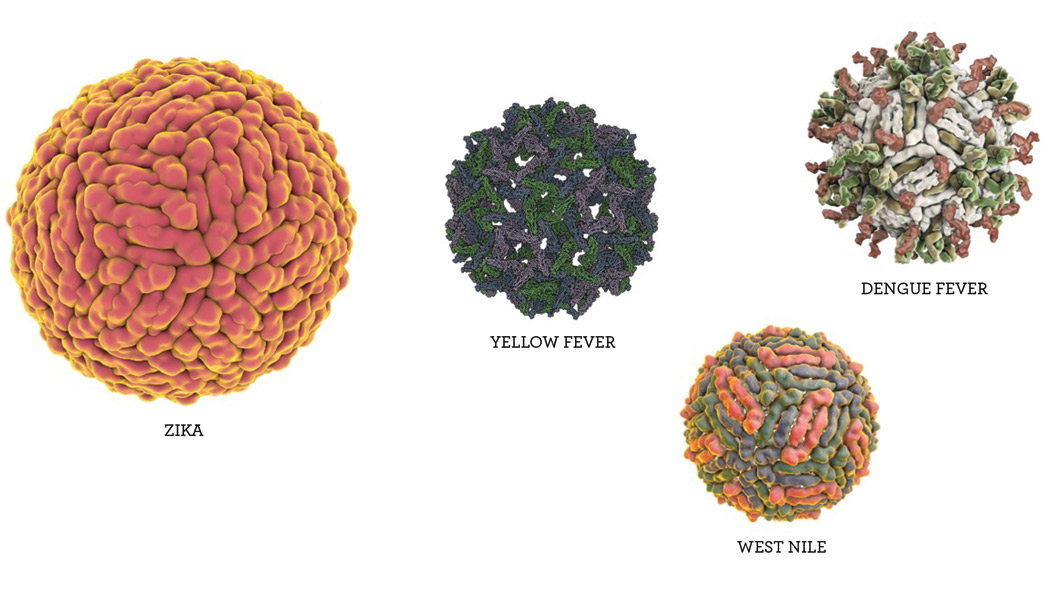 Zika’s extended family
Zika’s extended familyZika is actually a part of a notorious family of disease-causing pathogens called flaviviruses. Their name comes from the Latin “flavus,” which means yellow, a reference to the jaundiced skin and eyes of patients infected with yellow fever virus. Other members of the family include dengue virus, West Nile virus and Japanese encephalitis virus.
The good news about the relationships between these pathogens is that scientists have studied many of Zika’s cousins extensively, and the lessons they have learned may help speed discovery.
“Zika virus has a lot in common with other flaviviruses,” said Ross. “Vaccine candidates have been developed for dengue, West Nile and yellow fever, so that gives us encouragement that we can develop a vaccine for Zika virus as well.”
West Nile: Maintained in a natural cycle of transmission between birds and mosquitoes, West Nile virus can cause fatal neurological disease in humans, and it spread throughout much of the United States following the first reported cases in Queens, New York in 1999.
Yellow fever: Found in tropical and subtropical areas of South America and Africa, yellow fever generally only causes mild illness. However, approximately 15 percent of cases progress to a more severe form of the disease, which is characterized by high fever, jaundice, bleeding, shock and organ failure.
Dengue fever: Dengue fever is a leading cause of illness and death in tropical and subtropical regions throughout the world, infecting as many as 400 million people every year. In its most severe form, the infection causes blood vessels in the body to leak, which may lead to failure of the circulatory system.
Japanese encephalitis: While rare, fatality rates among those who develop encephalitis (an inflammation of the brain) from Japanese encephalitis virus can be as high as 30 percent. People who survive severe infections may experience permanent intellectual, behavioral or neurological problems, including seizures or paralysis.
Researchers at the University of Georgia and Sanofi Pasteur, the vaccines division of Sanofi, announced today the development of a vaccine that protects against multiple strains of both seasonal and pandemic H1N1 influenza in mouse models.
The University of Georgia announced today that it has entered into a collaborative research agreement with GeoVax Labs Inc. to develop and test a vaccine to prevent the emerging and virulent Zika virus infection.
By Stephanie Schupska
While scientists have known for years that African trypanosomes cause sleeping sickness, they’ve been left scratching their heads as to how these tiny single-celled organisms communicate. A University of Georgia study, published Jan. 14 in the journal Cell, helps solve this mystery.
The UGA researchers discovered that long filaments—that look like beads on a string—form by budding from the flagellum of African trypanosomes and then release pieces of the parasite into the host. This causes anemia and influences the outcome of infection leading to human African sleeping sickness and the cattle disease nagana.
The UGA researchers theorize that the extracellular vesicles, as the free-floating beads are scientifically known, are being used by the parasite to communicate with each other and with the host’s body. Even before they pop off into vesicles, the nanotubes extending from the flagellum help the single-celled parasites talk to each other. The severe anemia caused by the parasites may be an accidental side effect of the extracellular vesicles fusing with host red blood cells.
There were 6,314 new cases of African sleeping sickness in 2013. The disease, fatal if left untreated, threatens millions of people annually in the 36 countries in sub-Saharan Africa where the parasite-transmitting tsetse fly lives, according to the World Health Organization.
The research findings provide another clue to how African trypanosomes infect humans. It may also lead to improved therapies to fight sleeping sickness; current medications used to combat the disease have improved over the past decade but still include an old arsenic-based drug that kills between 5 and 10 percent of the people receiving treatment, said the study’s senior author Stephen Hajduk, a professor of biochemistry and molecular biology in the UGA Franklin College of Arts and Sciences.
The parasite also causes major economic losses by infecting and killing between 5 million and 7 million cattle each year through nagana, he said.
The research into trypanosome nanotubes and extracellular vesicles started as a side project in Hajduk’s lab about two years ago. As the study’s lead author Tony Szempruch spent more time peering into a microscope, the tiny, wiggly organism revealed its cellular communication potential.
“What you see here,” he said, pointing at the flagellum, “is that you can get that synthesis of the nanotube, but then it will quickly break down into what appears to be free vesicles that float out of focus.”
Szempruch, a doctoral student in the biochemistry and molecular biology department, developed a 3-D reconstruction of the nanotubes budding at the flagellum membrane. He was then able to look at the relationship of the flagellum, nanotubes and extracellular vesicles.
“The whole project developed out of our interest in how trypanosomes interact with one another,” Hajduk said. “Traditionally, people didn’t think of a single-celled organism needing to communicate with each other. But it has become more and more clear that they do.
“They’re actually able to sense when they’re at a certain level in the mammalian host in the bloodstream and then are able to respond to that in some way. As it turns out, a lot of this came together in looking at these extracellular vesicles that we’ve identified.”
Hajduk first noticed the nanotubes in 1978 when he was a doctoral student at the University of Glasgow, and they were first noted in a scientific publication in 1912.
“Even back then, we saw a lot of these extensions coming off the posterior end of the cell,” he said. “I think everyone has seen them, and, until now, everyone has ignored them. The parasite world—and trypanosome world—has largely lagged behind.”
Their findings—that nanotubes and vesicles are an important part of the communications process—show that the extracellular vesicles contribute to the complexity of African trypanosomiasis through the transfer of virulence factors between parasites and inadvertent interaction with host cells, which has a profound effect on disease, the study notes.
More research is needed into nanotubes in particular, Hajduk and Szempruch said. There’s also a great deal of interest in using the structures for non-invasive diagnostics and for targeted therapeutic use.
“The whole signaling thing, people are very excited about that,” Hajduk said, “whether it’s infectious disease or cancer or specific therapeutic development” to treat sleeping sickness.
“The fact that these vesicles are fusing with other host cells presents an interesting target for a therapeutic approach,” Szempruch said. “Perhaps treatment wouldn’t kill the parasite, but it would stop severe pathology associated with the parasite infection.”
UGA study co-authors included Steven Sykes, Rudo Kieft, Lauren Dennison, Allison Becker, Anzio Gartrell and William Martin, with John Harrington as a co-corresponding author, as well as Ernesto Nakayasu at Pacific Northwest National Laboratory and Igor Almeida at the University of Texas.
The study, “Extracellular vesicles from Trypanosoma brucei mediate virulence factor transfer and cause host anemia,” was supported by the National Institutes of Health under grant numbers AI039033, AI060546 and 2G12MD007592.

Stephen Hajduk is a professor of biochemistry and molecular biology in the Franklin College of Arts and Sciences.
By Beth Gavrilles
Researchers at the University of Georgia have developed a model for evaluating a potential new strategy in the fight against drug-resistant diseases.
The strategy would take advantage of parasite refugia—host populations that have not been treated with drugs, thereby serving as “safe zones” where parasites don’t develop drug resistance. When parasites from refugia mix with their drug-resistant counterparts in the general population, they could reduce the incidence of drug-resistance overall, which may help prolong a drug’s effectiveness.
The research, just published in the Royal Society journal Biology Letters, offers a way to assess when such an approach is likely to work—information that could help in the increasingly urgent hunt for alternatives to the current suite of parasite-fighting drugs.
“Once resistance emerges, you might squeeze a little bit of life out of a drug by tweaking it, but often, very quickly, that entire class of drugs will become useless,” said Andrew Park, an associate professor in the UGA Odum School of Ecology and College of Veterinary Medicine, who led the research.
“Right now, we’re really struggling to manage diseases like MRSA and extensively drug-resistant TB,” he said. “We’re at a point where new classes of drugs don’t grow on trees anymore; it can take 15 to 20 years to develop them. It’s a challenge just to keep pace. Refugia represent a management strategy that’s being considered as a way to slow down the development of drug resistance, particularly in animal health.”
Park and his colleagues created a model to predict the effects of refugia on two important outcomes: the overall prevalence of infection within a population and the proportion of those infections that are drug-resistant.
The model incorporates two main variables. One is the level of drug coverage—the proportion of the overall population that has been treated against parasites. The other is the degree of mixing between the treated and untreated groups.
In the case of a disease like heartworm, which is transmitted by mosquitoes and infects both wild animal populations and companion animals, there is some natural variation in both drug coverage and mixing. Both variables can also be controlled, to an extent.
“There might be variation in terms of how compliant animal owners are in giving heartworm preventive and whether dogs are kept indoors a lot versus just allowed to run around outside,” Park said.
In this case, the mixing is unintentional rather than planned. “Given that it’s happening, we should at least understand what the consequences might be for drug resistance,” he said.
The model yielded predictions about how changing the levels of drug coverage and mixing would affect prevalence and resistance, and also shed light on the evolutionary processes at work.
“The dogma is that increasing contact between treated and untreated groups would be predicted to simply increase prevalence and decrease frequency of resistance, but we found that the relationships were much more complex than that,” Park said. “And we gained a lot of insight into the different roles of selection—how different strains are favored because of the host environment—and gene flow, the movement of drug resistant and drug susceptible parasites between host groups.”
Park said that his team’s model serves as a general outline for considering the use of refugia as a management strategy, providing a blueprint for future models to predict outcomes in specific host-parasite systems.
“With refugia, the idea is to dilute overall drug resistance,” Park said. “We can say, yes, we’re putting parasites into the population, but they’re the right kind of parasites, and in some cases that may be better than having it just flooded with the wrong kind of parasites that we can never treat. But we need this kind of model to understand all these interactions in order to know when that’s a good idea.”
The paper’s coauthors are James Haven, a former postdoctoral associate at the Odum School; Ray Kaplan, a professor in the UGA College of Veterinary Medicine; and Sylvain Gandon of the Centre d’Ecologie Fonctionnelle et Evolutive in Montpellier, France.
The study, “Refugia and the evolutionary epidemiology of drug resistance,” is available athttp://rsbl.royalsocietypublishing.org/content/11/11/20150783.
By Molly Berg
A University of Georgia researcher has found that low levels of vitamin D may limit the effectiveness of HIV treatment in adults.
Those with human immunodeficiency virus—commonly known as HIV—often struggle with declining health because their immune systems can’t effectively respond to common pathogens. Their immune statuses, usually measured by CD4+T cells, normally improve when given HIV treatment.
“Because of the immune-destroying effects of HIV, infection usually results in relatively quick death without treatment. The magic of antiretroviral therapy, the name for drugs to treat HIV, lies in its ability to restore immune function,” said study co-author Amara Ezeamama, an assistant professor of epidemiology and biostatistics in the College of Public Health.
“With antiretroviral drugs, people with HIV are beginning to live longer lives. Our goal was to understand whether vitamin D deficiency limits the amount of immune recovery benefit for persons on HIV treatment.”
The findings were published recently in the journal Clinical Nutrition.
Ezeamama conducted an 18-month longitudinal study in which the immune status of 398 HIV-positive adults was measured at 0, 3, 6, 12 and 18 months. The researchers, through observation, related the rise in immune function to whether or not individuals had adequate levels of vitamin D.
Specifically, Ezeamama found that vitamin D helped the adults’ CD4+T cells recover more quickly. CD4+T cells are a type of T cell that helps the immune system fight off pathogens. For HIV-positive adults, CD4+T cells are critical because of their weakened immune systems.
Ezeamama found that participants with sufficient levels of vitamin D recovered more of their immune function—on average 65 CD4+T cells more—than those with vitamin D deficiency. The benefit of vitamin D sufficiency seemed greater for younger and underweight HIV-positive adults.
“HIV destroys the capacity of the body to mount effective response to pathogens,” Ezeamama said. “Given different vitamin D levels, HIV-positive adults recovered at different rates. We found a relationship between vitamin D and CD4+T cells.”
During the study, the participants were observed while on highly active antiretroviral therapy, or HAART, which normally consists of three or more drugs and is currently the most common—and most effective—treatment for HIV-positive adults. Those on HAART take it daily.
“It’s a cocktail of several medicines that controls the virus,” Ezeamama explained. “In addition to HAART, ensuring vitamin D sufficiency may also be helpful in restoring immune function.
“As researchers, we want to know what we can do to help. Vitamin D is relatively cheap. If we intervene with it, it could give individual HIV-infected persons a modest immune recovery bump that will likely translate to big public health impact.”
In the future, Ezeamama wants to look at how vitamin D affects immune recovery and long-term health outcomes in HIV-positive children.
“We are now in an era of hope for persons with HIV,” she said. “We know that HIV treatment works, and now people can live for several decades with HIV. We can further delay the progress of the disease and maintain survivors on a higher quality of life if we understand the factors that limit the effectiveness of HIV treatment.”
Study co-authors include David Guwatudde and Rachel Kyunene of Makerere University College of Health Sciences; Molin Wang, Wafaie W. Fawzi and Christopher Sudfeld of the Harvard University School of Public Health; Danstan Bagenda of Makerere University College of Health Sciences and Harvard School of Public Health; and Yukari C. Manabe of Makerere University College of Health Sciences and John Hopkins University.

Amara Ezeamama is an assistant professor of epidemiology and biostatistics in the College of Public Health.